


Study on the Alkali Corrosion Resistance of Aluminum Silicon Refractory Bricks Used in Cement Kilns
Time: 2025-03-28 11:38:06
Author: Zhengzhou Huaang Refractory Materials Co., Ltd
From: Find refractory materials network
Click:
1.1 Selection of refractory bricks
Choose commercially available silica brick 1680, silica brick 1550, anti stripping high alumina brick JA, and low alumina mullite brick M55. The physical and chemical properties of the sample bricks are shown in Table 1.
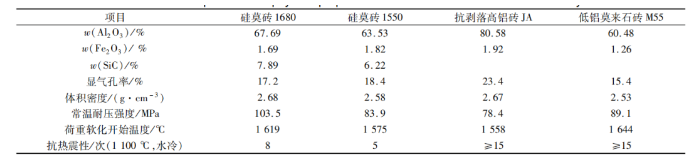
Table 1 Physical and Chemical Indicators of Commercial Aluminum Silicon Refractory Bricks
1.2 Alkali corrosion resistance test and performance evaluation
Conduct alkali corrosion resistance test using static crucible method. Cut sample blocks of size 80mm × 80mm × 80mm from various bricks, drill a cylindrical groove of 36mm × 40mm at the center to make a crucible, and then cut a thin plate of 60mm × 60mm × 30mm to make a crucible cover. Dry the crucible and crucible cover. Fill each crucible with 20g of commercially available chemically pure K? CO?, Seal the gap between the crucible cover and crucible with clay. The entire crucible is dried in an oven at 110 ℃ for 12 hours, then placed in an electric furnace and kept at 1100 ℃ for 5 hours until it cools naturally. Evaluate the alkali corrosion resistance of the sample by observing its appearance. Analyze the microstructure and phase composition of the corroded area at the bottom of the crucible. Divide the area by 5mm from the bottom of the crucible to the bottom of the brick sample (see Figure 1), and use EDS to analyze the K element content in each area. Use X-ray diffractometer to detect the phase composition of the corroded metamorphic layer in the 0-10mm area at the bottom of the crucible.

Figure 1 Schematic diagram of crucible sampling after alkali erosion
results and discussion
2.1 Appearance analysis of crucible alkali after erosion test
From the appearance photos of four types of crucibles after alkali erosion, it was found that there were no cracks in the silica brick 1680, silica brick 1550, and low alumina mullite brick M55, indicating excellent resistance to alkali erosion; However, the anti stripping high alumina brick JA has large penetrating cracks and relatively poor alkali resistance.
2.2 Distribution of K element at the bottom of the crucible after erosion
Perform EDS surface scanning on the bottom of the crucible adjacent to each partition line, and determine the K element content as shown in Figure 2. From Figure 2, it can be seen that the distribution of K elements varies greatly among different bricks. Among them, the variation curves of K element content in silica brick 1680 and silica brick 1550 are consistent, with the maximum K element content at 0mm; As the erosion depth increases, the content of K element decreases sharply. After reaching a depth of 20mm, the content of K element approaches 1% (w) and no longer changes. The distribution curves of K element content in anti stripping high alumina brick JA and low alumina mullite brick M55 are relatively similar. The anti stripping high alumina brick JA has the highest K element content at 0, 5, and 10mm; The highest point of K element content in low aluminum mullite brick M55 is at 0 and 5mm. After reaching the highest value of K element in these two types of bricks, the content of K element decreases sharply as it moves away from the bottom of the crucible hole; After 30mm, the potassium content of the anti stripping high alumina brick JA approaches 1% (w) and no longer changes; The potassium content of low alumina mullite brick M55 approaches 1% (w) after 20mm and no longer changes.
The alkali corrosion resistance of silica bricks is related to the introduction of silicon carbide and the apparent porosity of silica bricks. The silicon dioxide generated by the oxidation of silicon carbide at high temperatures reacts with potassium carbonate on the surface of refractory bricks to form a dense layer of glass phase, which can effectively suppress the erosion and infiltration of K elements, allowing K elements to concentrate in the surface area of the bricks. Anti stripping high alumina bricks and low alumina mullite bricks have high apparent porosity, which provides a fast permeation channel for molten potassium carbonate. Potassium carbonate enters the interior of the brick through the pores and reacts with the brick at high temperatures to form minerals such as garnet or white garnet. The formation of garnet or white garnet causes significant volume expansion, leading to cracking of the brick body. High porosity anti stripping high alumina bricks have higher K element content at the same depth compared to low porosity low alumina mullite bricks, and their K element enrichment is greater in the surface area.
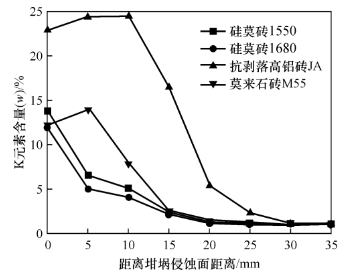
Figure 2 Changes in K element content at different locations in different areas of the bottom of different brick crucibles after alkali erosion
2.3 Analysis of microstructure and phase composition in the erosion layer of 0-10mm area
2.3.1 Silica brick 1680
Figure 3 shows SEM images of the erosion layer of 1680 silica bricks at different magnifications in the 0-10mm area after alkali erosion. Observing from Figure 3 (a), it can be observed that when in contact with K? The surface structure at the bottom of the CO crucible is dense, and cracks appear in the edge area of large particles. From Figure 3 (b), it can be observed that the edges of alumina particles in the matrix are significantly eroded, while there is no significant change in silicon carbide.

Figure 3: Microstructure photos of the 0-10mm area of the erosion layer on the 1680 silica brick at different magnifications
Figure 4 shows the XRD pattern of the erosion layer in the 0-10mm area of the 1680 silica brick. It can be seen that its phase composition is mainly composed of corundum, mullite, and silicon carbide, with K element present in the glass phase.
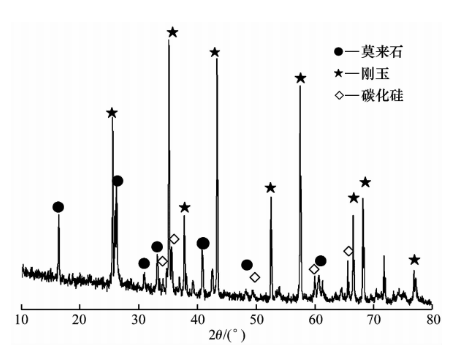
Figure 4 XRD pattern of erosion layer 0-10mm in 1680 silica brick
Figure 4 shows the reaction between alumina and silicon carbide in the Mo brick matrix, resulting in the formation of a liquid phase. The liquid phase fills the pores of the Mo brick and seals the pores and cracks in the contact area between the refractory brick and alkali, forming a dense layer that hinders further infiltration of K element.
2.3.2 Anti peeling high alumina brick JA
The microstructure photo of the 0-10mm area at the bottom of the JA crucible with anti stripping high alumina brick after alkali erosion is shown in Figure 5.
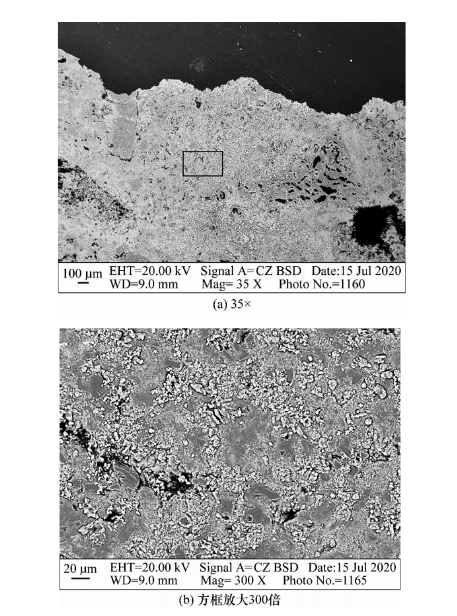
Figure 5: Microstructure photo of the 0-10mm area of the JA erosion layer on the anti stripping high alumina brick
From Figure 5 (a), it can be observed that the surface structure at the bottom of the crucible becomes denser, and the erosion of alumina particles is obvious, while the particle structure in the matrix becomes less obvious. From the observation in Figure 5 (b), it can be seen that the edges of the matrix particles become very blurry, and the pores or grain boundaries are filled with a large amount of light colored new phases. Anti stripping high alumina brick JA exhibits high porosity, and potassium carbonate enters the interior of the brick through pores or grain boundaries to corrode the sample, with a large depth of erosion. Through phase analysis of the erosion layer, it was found that potassium garnet was generated in the erosion layer (see Figure 6). The generation of a large amount of potassium garnet causes volume expansion, resulting in large through cracks in the anti stripping high alumina brick.
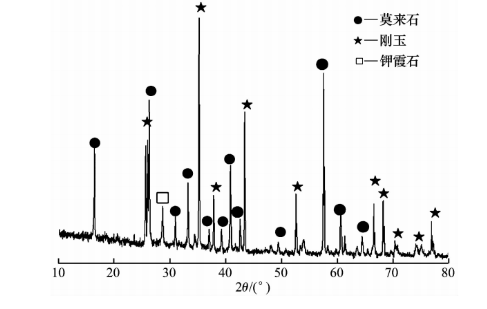
Figure 6 XRD pattern of JA erosion layer in the 0-10mm area of anti stripping high alumina brick
The microstructure photo of the 0-10mm area at the bottom of the low alumina mullite brick M55 crucible after alkali erosion is shown in Figure 7. From Figure 7 (a), it can be seen that there are also a few cracks on the surface structure of the bottom of the low alumina mullite brick M55 crucible, and the structure becomes denser, but there is a significant difference from the anti peeling high alumina brick. At high magnification (see Figure 7 (b)), the edges of dense mullite particles also show significant erosion, indicating that potassium carbonate also has a certain erosion and infiltration effect on low alumina mullite bricks, but it is significantly improved compared to anti stripping high alumina bricks. From the diffraction pattern (see Figure 8), it can be seen that the main phases of the erosion layer are mullite and a small amount of unconverted rhodochrosite, as well as a small amount of potassium garnet. The raw materials used in the low alumina mullite brick M55 are mullite homogeneous material and rhodochrosite. Mullite has a dense material structure and contains a certain amount of high silicon amorphous phase. At high temperatures, potassium dissolves into the amorphous phase to form a high viscosity glass phase; Meanwhile, the amorphous SiO formed by the decomposition of andalusite? Absorbing some potassium carbonate also generates a high viscosity glass phase, which seals the erosion layer of refractory bricks and hinders the penetration of K elements. A small amount of potassium carbonate reacts with the XRD pattern of the 0-10mm area of the erosion layer on the M55 alumina mullite brick to form potassium pyroxene. The amount of potassium pyroxene produced is low, and the resulting expansion is not sufficient to damage the brick structure. Therefore, the low alumina mullite brick M55 exhibits excellent resistance to alkali erosion.
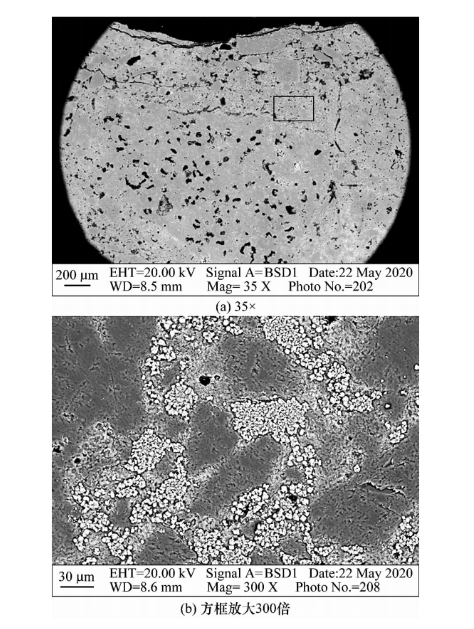
Figure 7: Microstructure photo of the erosion layer in the 0-10mm area of low alumina mullite brick M55
Conclusion
(1) The addition of silicon carbide and partial oxidation of silicon dioxide to fill the pores in silica bricks 1680 and 1550 can significantly reduce alkali corrosion and have excellent alkali corrosion resistance. After the alkali erosion test, the K element is mainly concentrated in the erosion surface area, and the content of K element decreases sharply with increasing distance.
(2) The commercially available anti stripping high alumina brick JA has a high apparent porosity and poor resistance to alkali erosion. The alkali erosion depth reaches 10mm, and the K element content (w) in the erosion layer reaches 20%~25%, generating a large amount of potassium garnet and causing the brick to crack.
(3) The low alumina mullite brick M55 has a low porosity, and the mullite material used has a dense structure compared to the andalusite material. A certain amount of high silica amorphous phase in the matrix can absorb alkali to form a high viscosity glass phase, which further seals the pores. The depth of alkali erosion is only 5mm, and the amount of potassium garnet generated by alkali erosion is relatively small, which is not enough to damage the brick structure. The resistance to alkali erosion is relatively good.






